Flow Cytometry & Cell Sorting Facility
About
The Naresh K. Vashisht College of Medicine Flow Cytometry & Cell Sorting Facility at Texas A&M Health Science Center provides cell analysis and cell sorting services for research in the Texas A&M community and is managed by the Department of Microbial Pathogenesis and Immunology.
The mission of the Fllow Cytometry and Cell Sorting Core is:
- To promote research and training for cell-based assays at the highest level of quality and rigor
- To bring new developments in flow cytometric instrumentation, techniques, and analysis for preclinical use in the Texas A&M research community
- To support the productivity and effectiveness of research by promoting high-content data acquisition, ensuring the highest data quality and consistency, carrying out efficient cell sorting, providing reliable data management, educating and training researchers, and consulting in experiment design and evaluation.
Major applications of flow cytometry include:
- Multi-parametric and high-resolution, phenotypic and functional characterization of immune and non-immune cells on a per-cell basis
- Cellular proliferation
- Stem, immune, and non-immune cell sorting to high-purity and viability
Instruments

Cytek Aurora 5L
- 5-laser configuration
- UV (355 nm)
- Violet (405 nm)
- Blue (488 nm)
- Yellow-Green (561 nm)
- Red (640 nm)
- 64 fluorescent detectors (16 UV, 16 Violet, 14 Blue, 10 Yellow-Green, and 8 Red)
- Equipped with optional plate loader

BD Fortessa X-20
- Violet (405)
- Blue (488)
- Yellow-Green (561)
- Red (640)
- 16 fluorescent channels
- Multi-parametric and high-resolution phenotypic and functional characterization on a per cell basis.
- Equipped with optional plate loader
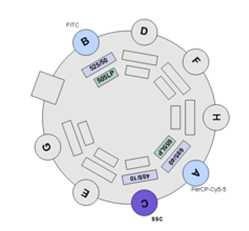
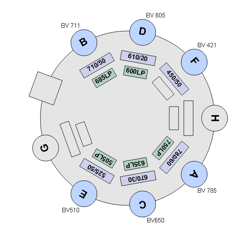
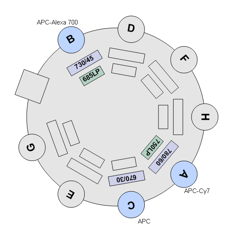
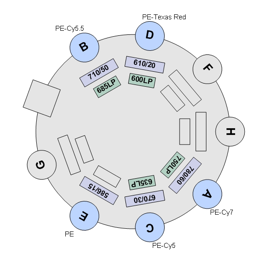

BDFACS Aria III
- Violet (405)
- Blue (488)
- Yellow-Green (561)
- Red (640)
- 13 fluorescent channels
- Up to 4-way bulk sorting
- ACDU attachment for sorting into well plates or slides
- Single Cell Index sorting

Miltenyi gentleMACS Octo Dissociator
- Semi-automated dissociation of tissues into single-cell suspensions or homogenates
- Optimized, pre-set programs
- Kits optimized for specific tissue types: mouse, human or rat-tumor, heart, neural tissue, spleen, lung, lamina propria, muscle, epidermis, liver and others

Seahorse XFe96 Analyzer
- 96-well plate format
- Measure OCR and ECAr of live cells
- Measure mitochondrial function
- Quantify ATP production
- Generate a metabolic phenotype within one hour
- Measure glycolytic rates in live cells
- Flexible assay design

Miltenyi MACSQuant Tyto Microfluidics Cell Sorter
- Three lasers (405, 488, and 638 nm) and 10-parameter cell sorting
- Alternative to droplet-based sorting
- Low Pressure (<3 PSI)
- Cells not subjected to electric charge
- No dilution of sorted fraction in buffer
- No aerosols generated
- Sterile, sealed cartridge
- Fast sorting with microchip valve that actuates 30,000 times per second

Curiox HT2000 96-Well Laminar Cell Washing System
- Alternative to centrifugal cell washing - better recovery
- Gentle on cells
- Greater reproducibility
Scheduling and Calendars
Before scheduling time in the Flow Cytometry and Cell Sorting Facility, you must obtain facility training. Please contact Robbie Moore, Flow Cytometry Specialist at rmoore@tamu.edu to schedule training.
In addition, prior to performing any work in the facility, compliance issues must be met. If your work in the facility requires IBC approval, you must add our facility to your protocol as a room amendment. You must also have your agents approved for use on our instruments. We require a copy of your IBC approval letter which states that you have express permission to run your agents on our instruments.”
All instrument scheduling is done through our iLab website. You must register for an iLab account before scheduling any instruments in the facility.
To register for an account:
To get started, you must register for an account:
- Navigate to the core page: https://tamu.corefacilities.org/service_center/show_external/4807
- In the upper-right-hand corner of the screen where it says, ‘Welcome TAMU user, please click here to login or register,’ select the click here link
- You will be directed to an authentication page where you will need to enter your TAMU credentials
- Once you have entered your credentials, click the ‘Login’ button
- You will be directed to an iLab Registration page where you will need to select your PI/Lab, and verify your contact information
- Once your registration has been submitted, your PI will receive a notification that you have requested membership to their lab in iLab. They will need to approve your membership and assign any TAMU Fund Name for your use.
To Create an Equipment Reservation:
Once you have been accepted into your PI’s lab and assigned TAMU Fund Name, you can schedule equipment time.
- Navigate to the core page: https://tamu.corefacilities.org/service_center/show_external/4807
- At the upper right hand of the page where it says, ‘Welcome TAMU user, click here to log in or register,’ select click here.
- Enter your TAMU Credential and password, and sign in.
- Select the Schedule Equipment tab and click on the ‘View Schedule’ button next to the instrument of interest. Click and drag on the time frame you would like to schedule your reservation for.
- A window will pop up that will allow you to verify your reservations details and provide payment information before saving the reservation.
Rate Structure
| Service | TAMU | External Academic | External Commercial |
| Aria Analysis | $51/hr | $76/hr | $102/hr |
| Aria Analysis w/ Operator | $55/hr | $83/hr | $110/hr |
| Aria Sort | $62/hr | $93/hr | $124/hr |
| Aria Sort w/ Operator | $70/hr | $105/hr | $140/hr |
| Seahorse | $18/hr | $27/hr | $26/hr |
| Fortessa Analysis | $49/hr | $73/hr | $98/hr |
| Fortessa Analysis Assisted |
$50/hr | $75/hr | $100/hr |
| Cytek Aurora Analysis | $62/hr | $93/hr | $124/hr |
| Cytek Analysis Assisted |
$50/hr | $75/hr | $100/hr |
| Curiox Cell Washer Use | $28/hr | $42/hr | $56/hr |
| MACSQuaint Tyto Sort | $61/hr | $91/hr | $122/hr |
| Assisted Instrument Use | $100/hr | $150/hr | $200/hr |
Training
- Before scheduling time in the Flow Cytometry & Cell Sorting Facility, you must obtain facility training.
- To schedule time for training on either the FACSAria, the Fortessa, the Cytek, or any other equipment, please send an email to Robbie Moore, Flow Cytometry Specialist,, at rmoore@tamu.edu
- Please indicate on which instrument you would like to receive training.
- Ms. Moore will confirm training availability and appointment date.
External Links
Fluorophore Spectrum Viewers:
Invitrogen:
Becton-Dickinson:
BD Fluorescence Spectrum Viewer
Flow Cytometry Tutorials:
Invitrogen:
Becton-Dickinson:
Resources and Vendors: